PART 2: What to Consider & How to Prepare for TCR studies
Considerations for IHC-based TCR Studies
According to FDA guidance in ‘‘Points to Consider in the Manufacture and Testing of Monoclonal Antibody Products for Human Use’’, frozen tissue is recommended for IHC based TCR studies. When IHC is used to perform TCR studies, the interaction of the antigen and the test biologic is key. The recognition of the epitope depends on the linear amino sequence and/or a three-dimensional conformation, which occurs when the protein is folded correctly. However, the fixation or freezing process of tissue can alter the protein folding, thereby affecting recognition of the epitope. For targets that are not proteins such as glycolipids, several fixation methods must be tested as stripping of epitopes can occur. Finally, the process of cutting tissue onto glass section can uncover intracellular epitopes that are not usually exposed in vivo, allowing the test biologic to bind. Therefore, what is observed on a glass slide may not necessarily reflect what occurs in vivo.
Another point to consider is that the test biologic may not be the most suitable reagent for IHC. A test biologic is developed because it has qualities that can influence the behavior of a biological process, and therefore, may not be the most optimal IHC reagent. Because regulatory agencies require that the test biologic be the main reagent in TCR studies, there are instances where the test biologic leads to less than ideal staining but still has potent biological activity.
Precursor Studies that Should be Performed Prior to TCR studies
GLP level TCR studies can be expensive and time consuming. It is highly recommended to perform several precursor studies to minimize delays in project timelines and accumulating costs. Prior to TCR studies, the target antigen-binding attributes of the biologic should be verified by comparing the binding by techniques such as surface plasmon resonance, flow cytometry, or ELISA. Under ideal circumstances, the binding of the antibody to the target should be tested with unlabeled antibody to confirm compatible antibody pairs for future IHC methods while using labeled antibody as a control. Target and antibody binding specificity within the disease context can sometimes be confirmed using peripheral blood mononuclear cells from normal or disease patients as well as other species such as non-human primate, rabbit, rat, and mouse. This is sometimes overlooked as the target expression profile may be differentially expressed (magnitude & distribution) in the normal versus diseased state.
Alternative approaches that can yield similar information as an IHC-based TCR study are tissue-based lysate western blots or flow cytometry with cell lines that express the target antigen. Tissue lysates can typically be purchased from PromoKine and cell lines from ATCC or DSMZ. In some cases, unique target antigen expression can be engineered into certain cell lines creating a positive (transduced/transfected) cell line and a negative control cell line (parental/mock transfected). These can be used for both flow cytometry and can be paraffin embedded for IHC methods development purposes. Finally, some preclinical groups are also using in-silico modeling to understand the potential for cross-reactivity that may predict possible antibody:epitope interactions not previously anticipated.
Benefits and Limitations of IHC-based TCR studies
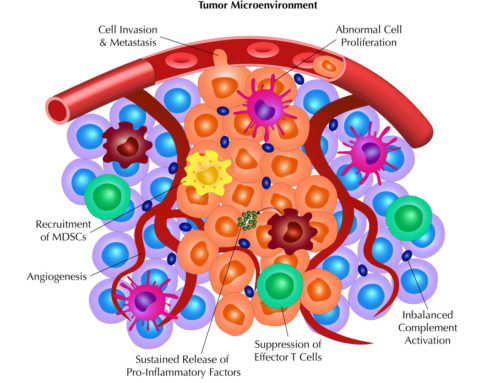
There are many benefits from screening the test biologic over various tissues of human or non-human origins. It can confirm that the target tissue is indeed expressing the epitope, which can then assist in the designing of in vivo toxicity experiments. TCR studies can also alert investigators to potential off-target tissues binding of the test biologic, which would make them aware of potential toxicity issues that could be observed in human clinical trials. Meanwhile, data from staining of tissues from different non-human species can direct the investigators towards the correct animal species to perform preclinical experiments that are most relevant to human. Finally, TCR studies can add more value to a test biologic by expanding the target antigen expression profile that can be followed up by diseased tissue arrays such as tumor microarrays for oncology indications.
While there are important benefits to IHC-based TCR studies, there are limitations as well. There are instances where a robust TCR assay cannot be developed; again, highlighting the fact that the test biologic may not be the most optimal IHC reagent. Next, there are studies that show no in vivo toxicity effects in light of positive tissue staining. In contrast, there are cases of in vivo toxicity even though the tissue did not stain positively. Together, these observations suggest that TCR studies be used as a screen in conjunction with other data to determine the best course of study for the test biologic. For example studies that may be relevant to your therapeutic, Leach et al (2010, Toxicologic Pathology) has published several case studies from various pharmaceutical and biotech companies to illustrate encountered scenarios and clinical findings.
Conclusion
While TCR studies provide many benefits, they are best used as supplementary data with other studies. Because of technical difficulties or non-optimal staining conditions and reagents, there are many cases where data from TCR studies can be misleading. Performed along with other types of experiments, TCR studies can provide guidance for suitable in vivo and follow up experiments to make inform decisions on the direction of test biologics. Thus, data from TCR studies should be carefully interpreted and in the context of other data on a case-by-case basis.
(If you missed the first part of this series: PART 1: Why Drug Development Values TCR Studies)