What is CDC and why would you need it?
This is Part 2 of our series, “The Power of Complements…”. Miss Part 1? Catch up here.
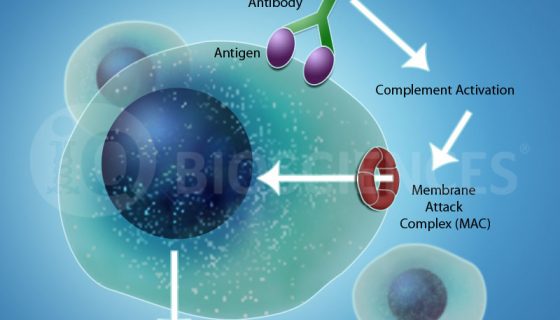
Potential applications & implications for CDC assays
As alluded to in Part 1, one of the most important applications for CDC assays is to determine if a potential therapeutic antibody can mediate CDC activity. Originally thought to only prevent downstream signaling of the target receptor by blocking its oligomerization or engagement to ligands, most therapeutic antibodies are now suggested to work through multiple mechanisms, including CDC activity. Thus, many companies screen their therapeutic antibody candidates for CDC activity to find ones that may be most efficacious due to their ability to mediate multiple mechanisms of action.
Another significant application for CDC assays is to assess the biopotency of approved drugs before they are released to hospitals and clinics. In compliance with regulatory agency guidelines, each lot of antibody that is produced must be tested for efficacy and have consistent IC50 within predefined specifications. This ensures consistency between drug batches, in order to prevent variations in efficacy once administered to patients.
CDC activity also has relevant implications for biomarker development. In this case, the cleaved complement products can be measured in the blood of individuals receiving certain therapeutic antibodies to assess the level of complement-mediated activity. The data can then be correlated with efficacy to provide beneficial information that can be used to identify individuals that will benefit most from the therapeutic. Thus, designing a diagnostic to measure cleaved complement components can be of great value, and understanding whether or not a therapeutic antibody has CDC activity is quite meaningful.
Finally, biopharmaceutical molecules or medical devices can also cause off-target complement activation that leads to destructive inflammation. Unlike IgE-mediated allergy, complement-mediated reactions can occur at the first exposure to a drug without prior sensitization. Thus, CDC assays are critical in testing for these unwanted side effects to prevent any complications upon application.
How are CDC assays performed?
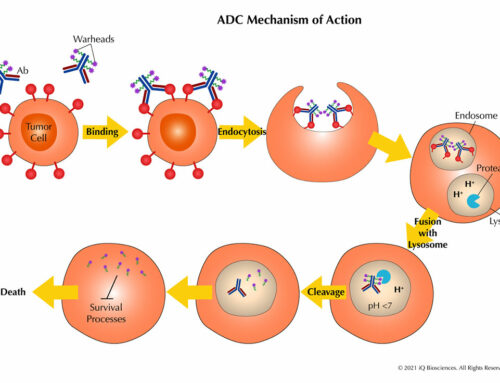
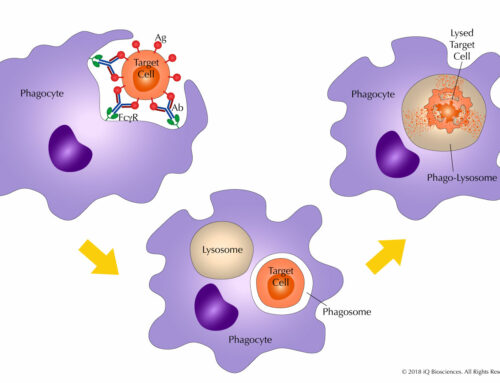
The main components for a CDC assay are: (1) the antibody, (2) target cells that express the antigen for the aforementioned antibody, and (3) serum. Once combined, the reaction is incubated for complement system activation and target cell lysis.
Target cell lysis can be measured in a multitude of ways. Traditionally, target cells were loaded with radioactive chromium, and then radioactivity in the supernatant was measured after the reaction. However, newer and safer methods have been developed, including measurement of lactate dehydrogenase (LDH), which is given off by lysed cells, in the supernatant. Yet another method is to measure the loss of target cells by flow cytometry; in this case, target cells are loaded with a fluorescent dye prior to incubation with the antibody and serum.
Most CROs with experience performing CDC assays prefer the latter two methods due to safety concerns and assay sensitivity. More so, certain CROs, such as iQ Biosciences, will work directly with clients to design assays based on their needs and have a deep knowledge of CDC assays to properly design these studies.
Considerations for performing CDC assays
Although the procedure to perform CDC assays seems fairly straightforward, there are several factors to consider for optimal assay performance. First and foremost is the type of serum used in the experiment: the serum must be from a good source to ensure purity and complement protein integrity. Second, the species of serum complement used in the experimental system is extremely important for providing physiologically relevant data. Third, the target cell line should express enough of the antigen so that the antibodies can form a complex with them. In addition, knowing the expression level of inhibitory receptors, such as CD46, CD55 and CD59, on the surface of target cells is important for interpreting data and designing future in vivo experiments. Finally, the assay used to measure cell death can be significant. Measurement of molecules in the supernatant is an indirect method of assessing cell death and can lack the sensitivity for detecting subtle amounts of killing. If a sensitive method is required, measurement of cell death by flow cytometry may be the ideal choice.
When selecting a CRO to perform CDC assays, researchers should look for those that have extensive knowledge and experience performing CDC assays, such as iQ Biosciences. Having these qualities will ensure that your experiments are optimally designed to obtain the most physiologically relevant data.
Learn more about iQ Biosciences‘ expertise in performing CDC assays.