With the current trend of developing immunotherapies for cancer, many of these therapies center on exploiting the potent effector functions of lymphocytes, specifically T cells. In our previous blogs, we introduced a new class of therapeutic molecules called bi-specific antibodies and discussed their mechanism of action for anti-cancer therapy. In this blog, we will discuss in vitro assays that use T cells to develop bi-specific antibody programs.
What are CD4 and CD8 T cells?
In most organismal immune systems, there are two main types of T cells. One type is the CD4 T cell, which is a hematopoietic cell that develops in the thymus and subsequently activates the immune system to mount an immune response. This cell becomes activated after binding the target cell, which then leads to the secretion of numerous cytokines that activate and potentiate the immune response. These cytokines include IFN-g, TNF-a, IL-2, IL-12, and IL-23.
CD4 cells are characterized by the presence of the CD4 marker and CD3/T cell receptor (TCR) complex on the surface of the cells. In a normal scenario, a T cell begins to secrete cytokines after engagement of its CD3/TCR complex to MHC class I protein on a target cell.
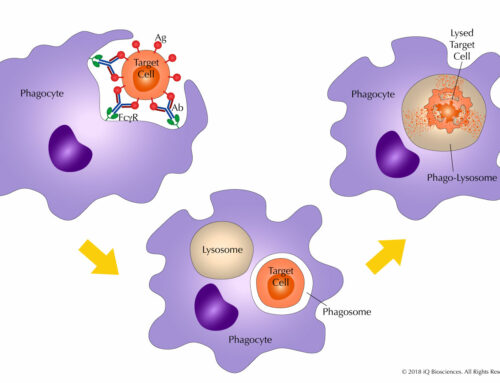
The other main type of T cell is the CD8 T cell, which also develops in the thymus. In contrast to mature CD4 cells, CD8 cells survey the body for aberrant cells, such as those that are infected or cancerous, to eliminate them. These cells perform their effector function by binding the target cell and secreting cytolytic molecules toward it, which leads to cell death. These molecules, which include perforin and granzyme, create holes in the target cells to permit entry of proteases that promote programmed cell death.
CD8 T cells can be identified by the presence of the CD8 marker and CD3/T cell receptor (TCR) complex on the surface of the cells. These cells are typically activated to kill upon engagement of their CD3/TCR complex to the MHC class I protein presenting foreign antigen on the target cells.
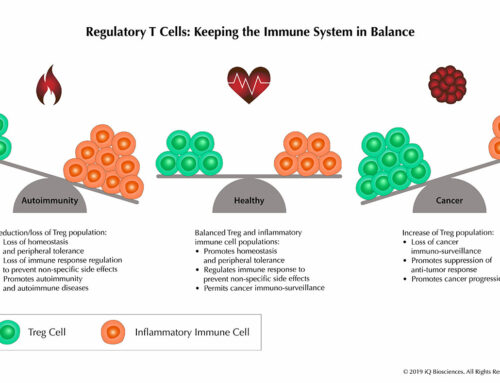
Because of these abilities to promote the immune response and perform cytotoxicity to eliminate aberrant cells, T cells are often exploited in cancer immunotherapies, such as bi-specific antibodies, checkpoint inhibitors, and genetically engineered T cells (CAR T cells). Therefore, T cells are important components in the testing and development of these therapies.
What are some T cell-based in vitro assays to assess bi-specific antibody specificity and function?
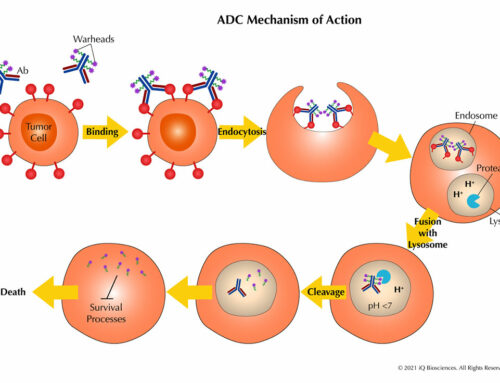
Bi-specific antibodies that exploit T cells often bring the T cell into close proximity with the target cell. Therefore, one arm of the bi-specific antibody must bind a marker expressed on the T cell surface, which in most cases is the CD8 or CD3 molecule. For development of these bi-specific antibodies, one type of required assay is the T cell binding assay to ensure the bi-specific antibody is binding the correct cell type. Typically, the assay is flow cytometry-based and performed with PBMCs, an assortment of cell-specific antibodies, and the bi-specific antibody. The data are then analyzed to determine the cell type that binds the bi-specific antibody, which should be T cells. Therefore, both CD4 and CD8 T cells are used for this binding assay.
Another in vitro assay commonly used in preclinical studies are functional studies that determine if the CD8 T cells are killing their targets. These cytolytic cells are mixed with targets in the presence of the bi-specific antibody and then assessed for cell death of the targets using various readouts. One method for assaying cytotoxicity is labeling target cells with Calcein, which is released upon cell death, and then measuring the amount of Calcein present in the supernatant of the culture. Another method uses flow cytometry. In this assay, the targets are also labeled with an intracellular dye and then assessed for death by the loss of the labeled target cells in the culture.
To focus on CD4 T cell function and bi-specific antibodies, one commonly performed assay is the cytokine release assay (CRA). These studies assess whether the antibody activates CD4 cells to promote cytokine secretion. Here, CD4 cells are mixed with targets in the presence of the bi-specific antibody and then the supernatant is collected and assessed for the presence of cytokines using a cytokine bead array (CBA) kit. This kit permits the interrogation of a number of different cytokines that may be produced.
With these T cell-based assays, investigators can perform pre-clinical studies to investigate the specificity and function of T cells in relation to bi-specific antibodies. Immunology-based CROs, such as iQ Biosciences, can assist researchers in determining which T cell types and assays will help in development of their bi-specific antibody. Contact iQ Biosciences for more information.
In our next blog, we will discuss T cell-based in vivo assays to assess the efficacy of bi-specific antibodies. Furthermore, we will review in vitro and in vivo data from some of our studies that put these T cell-based assays into context.