- Evaluate your immune checkpoint (CTLA-4, PD-1, TIM-3, LAG-3, TIGIT, OX40, GITR, etc.) biologic or molecule for specific binding to the receptor using ELISA or iQ’s cell line-based binding assay
- Binding affinities (EC50s) or binding inhibition (IC50s) can be generated with your molecules
- Personalized experimental design to ensure the most relevant data is obtained
- All assays are performed with high-quality reagents and strict controls by experienced immunologists
Service Overview
About the Immune Checkpoint Binding and Interaction Assays
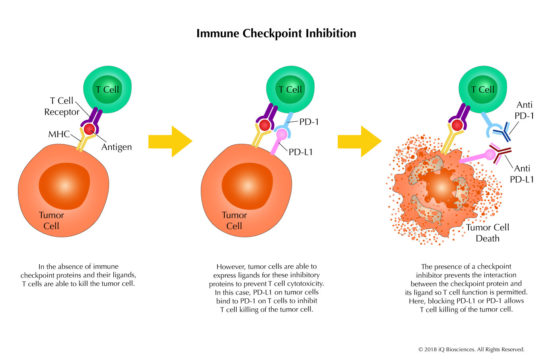
Immune checkpoint proteins are surface molecules on immune effector cell populations that inhibit effector function when engaged to their cognate ligand(s). Expression of these ligands on cancer cells has been suggested as a mechanism by which these cells evade the immune response. Thus, therapeutics called immune checkpoint inhibitors that block interaction between the immune checkpoint protein and their ligands can restore the effector function of immune cells and promote tumor regression.
iQ’s Biosciences’ Immune Checkpoint Assays
iQ Biosciences is an immunology CRO with years of experience helping companies develop their immune checkpoint inhibitors, whether they are antibodies, peptides, or small molecules, using our immuno-oncology assays. We can help design a roadmap that begins with binding assays to screen for good candidates, proceeds to interaction assays with selected candidates for blocking ability, then progresses finally to functional assays that test for immuno-modulatory activity. See our Checkpoint Inhibitor Functional Assay for more information about functional assays. With this roadmap, we can demonstrate the binding, interaction, and functional properties of your molecules.
Discover Your Molecules’ Binding Properties with iQ Biosciences’ Immune Checkpoint Binding and Interaction Assays
At the beginning of that roadmap, initial characterization of the checkpoint inhibitor is required to ensure its specificity for the immune checkpoint protein or its ligand, which can be accomplished using iQ Bioscience’s Immune Checkpoint Binding Assays. For these assays, we use a flow cytometry-based method to demonstrate binding of candidate molecules to cell lines expressing the protein of interest and to ensure specificity using cell lines that do not. Further evaluation of selected candidates can be tested for EC50 using this assay as having a selection of candidates with a wide range of EC50 is important. In some cases, candidates with the best EC50 are not necessarily the ones that move forward for development. See Figure 1 for an example of our Immune checkpoint Binding assay by flow cytometry.
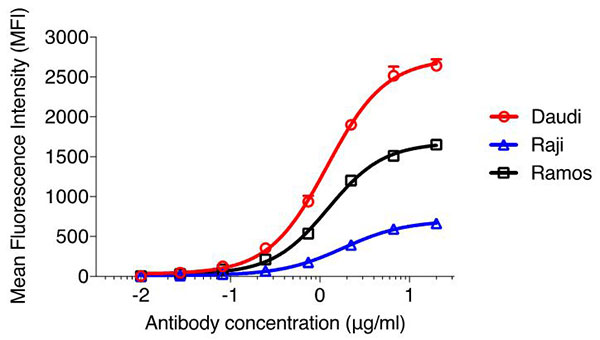
Figure 1. EC50 binding curves of CD38 antibody binding to cell lines expressing different levels of CD38. Daudi, Raji, and Ramos cells were stained with increasing concentration of the same fluorescently-labeled CD38 antibody. Flow cytometric analysis was performed on the cells and the mean fluorescence intensity (MFI) was obtained. Graphpad Prism was used to generate the binding curve of antibody concentration versus MFI.
After initial characterization to establish specificity of your candidate checkpoint inhibitors, experiments must be performed to demonstrate it can block interaction between the checkpoint protein and its ligand. Without the ability to block engagement, the candidate is impractical as engagement will likely lead to lymphocyte inactivation. Indeed, the mechanism of action in current FDA approved checkpoint inhibitors is thought to be interference of binding between the immune checkpoint and its ligand. However, when screening antibodies that bind, there may be candidates that recognize the target, but do not prevent interaction with its ligand. Therefore, interaction assays must be performed to ensure the checkpoint inhibitor prevents engagement, which is an important factor to understand for a functional immune checkpoint inhibitor. See our Checkpoint Inhibitor Functional Assay for other factors to understand. At iQ Biosciences, we have developed custom Immune Checkpoint Interaction Assays using a flow cytometry-based method to screen for potential candidates that interfere with binding. In addition, these studies suggest a potential mechanism of action for the checkpoint inhibitor if immune response function is restored.
iQ Biosciences – Your Immunology CRO for Checkpoint Inhibitor Development
In conjunction with our immune checkpoint inhibitor functional assays, we can demonstrate the binding and interaction properties of your molecule(s) . iQ’s personalized service will work with you to design a roadmap to determine the best assays to fulfill your program’s scientific requirements and to ensure you receive the most relevant data. We are your immuno-oncology CRO to bring that roadmap into focus and to execute its studies. Please contact us for more information.