- Examine ADCC activity of your therapeutic antibody with iQ’s antibody-dependent cellular cytotoxicity assay (ADCC assay)
- We utilize two different approaches to evaluate ADCC activity:
- ADCC reporter assays using cell lines that have either the V Variant or F Variant of position 158 of the FcgRIIIa receptor
- ADCC assays using primary cells (PBMCs, isolated NK cells) that have been characterized for V Variant or F Variant of position 158 of the FcgRIIIa receptor
- Personalized experimental design to ensure the most biologically relevant data is obtained
- Assays can be performed with purified effector cells or PBMCs
- All assays are performed with high-quality reagents and strict controls by experienced immunologists
Service Overview
About Antibody-Dependent Cellular Cytotoxicity (ADCC)
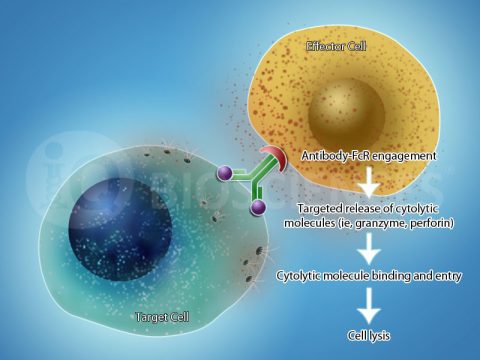
Antibody-dependent cellular cytotoxicity (ADCC), also referred to as antibody-dependent cell-mediated cytotoxicity, is a cytolytic function by which effector cells with cytotoxic potential, such as Natural Killer cells, lyse target cells. This function is dependent on an antibody, which serves as a bridge between the target cell and the effector cell. Mechanistically, the antibody binds its cognate antigen on the target cell through its antigen recognition domain, and then recruits the effector cell to the target with its Fc region. Once bound, the effector cell is in proximity of the target cell and can secrete cytolytic molecules to kill it.
ADCC can play roles in general human health and mechanism of action for therapeutic antibodies. First, it eliminates foreign pathogens and aberrant cells that would otherwise promote illness in general human health. Second, it is suggested that ADCC is a mechanism of action for many therapeutic antibodies, such as those that target HER2, CD20, and CD52. During drug development, potential therapeutic antibody candidates will be screened for ADCC activity to find ones that will have the most potent anti-cancer abilities.
iQ’s ADCC Assays: Lab-Tested, Immunologist-Approved
iQ has extensive experience with ADCC assays and can help you design the best experiment, keeping in mind factors that influence ADCC to determine the ADCC potential of your therapeutic antibodies. Because there are multiple components to ADCC, three well-studied factors can enhance or decrease ADCC:
- Glycosylation state – antibodies are glycosylated and the oligosaccharides expressed on them can affect ADCC
- FcgRIIIa (CD16) polymorphism – the FcgRIIIa binds the Fc region of the antibody, which comes in two polymorphisms wherein the hi-affinity valine form binds stronger than the low-affinity phenylalanine form and leads to more potent ADCC
- Expression level of antigen on target cells – the target cell line should express sufficient target antigen for the antibodies to form immune complexes so activation signals can be elicited to drive ADCC.
At iQ, we have moved away from traditional cytotoxicity assays that typically load target cells with radioactive chromium, which is released upon lysis. Instead, we primarily perform ADCC assays by high-throughput flow cytometry, wherein target cells are labeled with a fluorescent dye, to differentiate the targets from effector cells. After the target and effector cell co-culture, samples are analyzed with the flow cytometer along with a viability dye to assess the loss of membrane integrity of target cells in the culture, as a measure of cytotoxicity.
An alternative method that we employ at iQ is to use genetically engineered effector reporter cell lines that express luciferase upon FcgRIIIa binding to antibody. With this method, luciferase expression is measured instead of cell death as these effector cells are non-cytotoxic. This method provides the advantage of using a stable, reproducible effector cell line as compared to human effector cells, which can vary in activity from donor to donor.
With our extensive knowledge of factors that influence ADCC and our wide array of readouts for ADCC, iQ can help design the right experiments for you to determine the ADCC potential of your therapeutic antibodies.
Request an ADCC Fact Sheet
Interested in learning more about our ADCC Bioservices? Complete the form below to request a Fact Sheet.
