We Are Here to Help
Drug development in the research and preclinical stages is tricky to navigate and riddled with potential pitfalls. Our team of experienced immunologists has been down that road and intimately understands the process and the rigor involved. As veterans in the field, we want to impart that knowledge onto you to help guide your drug development efforts.
“Does my therapeutic modality bind?”
Your molecule won’t be effective unless it binds to the target of interest. We provide a number of assays to verify binding and specificity for your antibody or CAR-T cell therapeutic.
“Does my therapeutic perform the expected activity?”
Need to confirm the mechanism of action of your therapeutic antibody? Is it ADCC, CDC, ADCP, or multiple mechanisms? We specialize in designing and executing assays to determine this.
“Is my therapeutic safe?”
You may determine that your therapeutic functions as desired during early drug development, but it is absolutely critical to rule out the potential for unwanted side effects, such as an infusion reaction or cytokine release, prior to clinical trials. We have the expertise to design these types of assays.
Who Have We Helped?
Pharmaceutical
Biotechnology
Diagnostic
Academic
A Step Apart from Other Immunology-Based CROs
It’s in our DNA to deliver comprehensive end-to-end service to our customers. We understand the pain points customers have faced working with other CROs and we proactively tackle these challenges throughout our engagements to give customers confidence they’ve made the right choice to work with us.
We Listen
Active listening is critical to understanding our client’s projects and goals, so we give you our undivided attention during introduction calls to gather vital details that will impact your study design.
We Engage
The success of your project means a lot to us, so we ask relevant questions and do our homework based on your inputs to provide you with meaningful and detailed feedback on your study objectives.
We Guide
Nobody likes making known mistakes, especially when time and money are on the line. Therefore, our immunology specialists help guide you down the right path based on their experience and don’t sell you on extras you don’t need.
We Communicate
We know customers don’t like to be kept in the dark on projects that are near and dear to their hearts. So during project engagements, we keep you apprised of progress and provide a heads up on any warning signs.
We Manage
Building a plan. Tracking to timelines. Raising risks and issues. Ensuring committed resources are on point. We believe all that is Project Management 101, and that’s how we will manage your projects.
We Share
To us, it’s not just about giving you a data dump of your project results and letting you go on your merry way. We perform critical analysis of your results and share our key insights with you to help you establish next steps.
Generated Annually
Experimental Results
Immunology Experience
Getting Started on a Project is as Simple as 1-2-3
Schedule an appointment
Through an introductory call with one of our immunology experts, we’ll discuss the parameters and goals of your prospective project and provide any insights we have based on our experience. This will set the stage for the study design.
We’ll create a plan
Based on your study parameters and the project goals discussed, we’ll put together a study design in the form of a detailed project proposal and send it your way to review and approve.
Remember, customized quotes are FREE!
We’ll execute the plan, together
Once you’ve given us the green light on the project proposal, we can get to work right away. We stay on top of obtaining the necessary reagents and/or test articles to complete your project in a timely manner.
Don’t fret! We’ll keep you informed on your project’s progress every step of the way.
iQ Learning Series
We endeavor to educate.
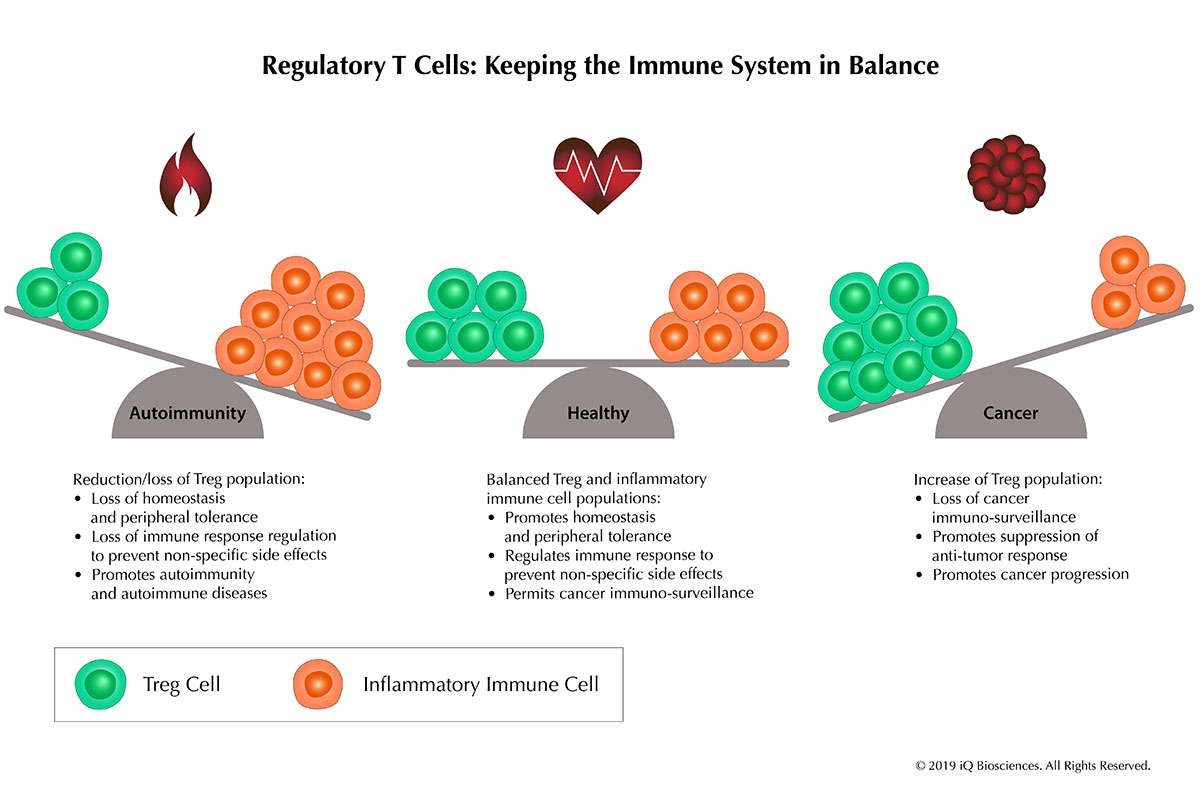
The Suppressive Nature of Regulatory T Cells and Their Roles in Cancer and Diseases
Introduction to Regulatory T Cells The immune response is an exquisitely controlled event in which lymphocytes expand, secrete cytokines, and kill infected cells in [...]
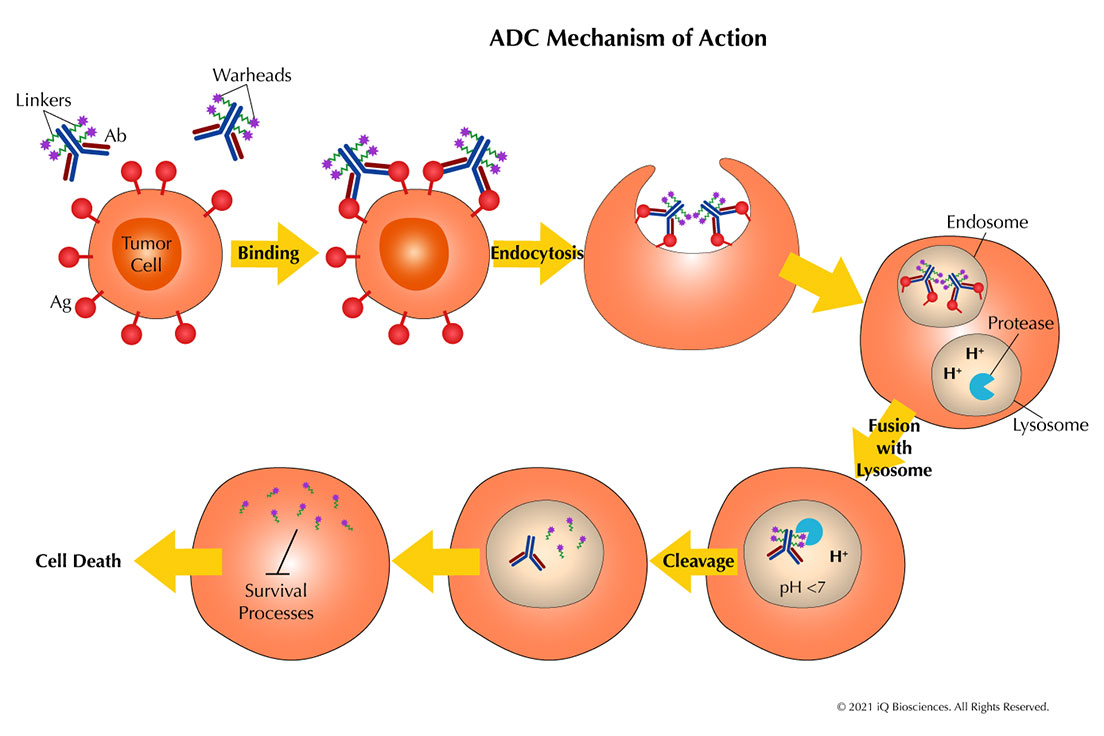
The ABCs of ADCs: An Introduction to Antibody-drug Conjugates
The terms “warhead” and “payload” typically invoke impressions of weapons and destruction. However, in a more constructive and positive light, these terms are now [...]
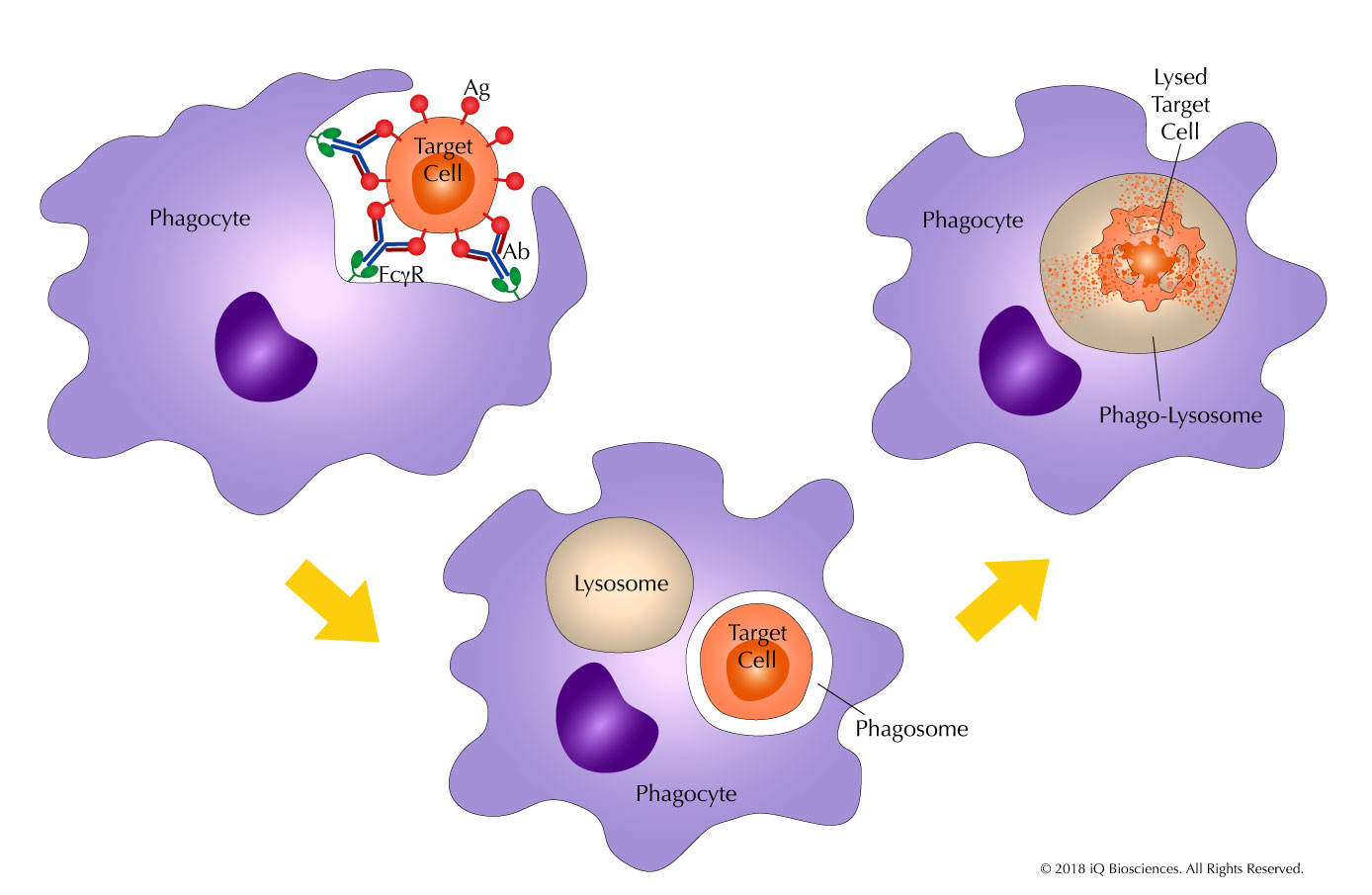
ADCP: The Covert MoA for Therapeutic Antibodies You Need to Know About (Part 2)
In our previous blog, we discussed antibody-dependent cellular phagocytosis (ADCP), a mechanism of action for therapeutic antibodies that is becoming increasingly appreciated. In [...]